ThermodynamicsHard
Question
A monoatomic ideal gas, initially at temperature T1, is enclosed in a cylinder fitted with a frictionless piston. The gas is allowed to expend adiabatically to a temperature T2 by releasing the piston suddenly. If L1 and L2 are lengths of the gas column before and after expansion respectively, then T1 / T2 is given by
Options
A.(L1 / L2)2/3
B.(L1 / L2)
C.(L2 / L1)
D.(L2 / L1)2/3
Solution
During adiabatic expansion, we know
TVγ-1 = constant
or T1V1γ-1 = T2V2γ-1
For noatomic gas, γ = 5 / 3
∴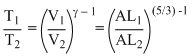
[A = area of cross section of piston]
= (L2 / L1)2/3
TVγ-1 = constant
or T1V1γ-1 = T2V2γ-1
For noatomic gas, γ = 5 / 3
∴
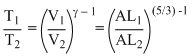
[A = area of cross section of piston]
= (L2 / L1)2/3
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
A black body, at a temperature of 727oC, radiates heat at a rate of 20 cal m-2s-1. When its temperature is raised to 727...A gas is found to obey P2V = constant. The initial temperature and volume of gas are T0 & V0. If the gas expands to volu...The volume of a 2 mole ideal gas at 27oC is V. It expands adiabatically to the volume 2V. If γ = 5/3, R = 8.31 J/mo...Two rigid boxes containing different ideal gases are placed on a table. Box A contains one mole of nitrogen at temperatu...At a constant temperature and volume for a given gas, pressure...