ThermodynamicsHard
Question
Two rigid boxes containing different ideal gases are placed on a table. Box A contains one mole of nitrogen at temperature T0, while Box B contains one mole of helium at temperature (7/3) T0. The boxes are then put into thermal contact with each other and heat flows between them until the gases reach a common final temperature. (Ignore the heat capacity of boxes). Then, the final temperature of the gases, Tf, in terms of T0 is
Options
A.Tf = 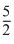 T0
T0
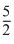 T0
T0B.Tf = 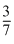 T0
T0
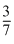 T0
T0C.Tf = 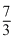 T0
T0
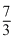 T0
T0D.Tf = 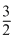 T0
T0
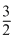 T0
T0Solution
ᐃU = 0
⇒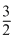 R(Tf - T0) + 1 ×
R(Tf - T0) + 1 × 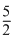 R(Tf -
R(Tf - 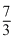 T0) = 0
T0) = 0
Tf =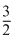 T0
T0
⇒
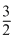 R(Tf - T0) + 1 ×
R(Tf - T0) + 1 × 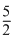 R(Tf -
R(Tf - 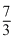 T0) = 0
T0) = 0Tf =
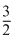 T0
T0Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
As shown in the diagram for a closed path ABCA :...Heat travels through vacuum by :...Water is used to cool the radiators of engines in cars because :...The process in which the heat given to a system is completely transformed into work is for ddeal gas : -...Helium gas goes through a cycle ABCDA(consisting of two isochoric and two isobaric lines) as shown in figure. Efficiency...