ThermodynamicsHard
Question
A gas is found to obey P2V = constant. The initial temperature and volume of gas are T0 & V0. If the gas expands to volume 3V0. Find the final temperature of gas.
Options
A.T0
B.√3T0
C.2T0
D.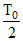
Solution
P2V = constant
also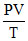 = constant ⇒
= constant ⇒ 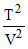 V = constant
V = constant
∴ T2V-1 = constant
∴ Tf2 = T02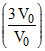 ⇒ Tf = √3T0.
⇒ Tf = √3T0.
also
∴ T2V-1 = constant
∴ Tf2 = T02
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Specific heat of a gas undergoing adiabatic change is :...During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its absolute temperature. ...When you make ice cubes, the entropy of water...What is the amount of work done, in giving . 400 calories of heat to a system :-...A shown in the figure, the amount of heat absorbed along the path ABC is 90J and the amount of work done by the system i...