ThermodynamicsHard
Question
The temperature of an ideal gas is increased from 120 K to 480 K. If at 120 K theroot mean square velocity of the gas molecules is v, at 480 K it becomes
Options
A.4 v
B.2 v
C.v / 2
D.v / 4
Solution
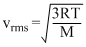
i.e., vrms ∝ √T
When temperature is increased from 120 K to 480 K (i.e., four times), the root mean square speed will become √4 to 2 times i.e., 2 v.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
A black body has maximum wavelength λm at 2000 K. Its corresponding wavelength at 3000 K is:...A refrigerator with coefficient of performance 1/3 releases 200 J of heat to a hot reservoir, then the work done on the ...If the quantity of energy with drawn from a system is δQ, work done by the system δW and the increase in inter...An ideal gas is subjected to an isothermal expansion such that its volume change fromVi to Vf and pressure from Pi to Pf...A gas is taken through the cycle A → B → C → A, as shown, What is the net work done by the gas ?...