ThermodynamicsHard
Question
An ideal gas is subjected to an isothermal expansion such that its volume change fromVi to Vf and pressure from Pi to Pf The work done on the gas is :
Options
A.W = + nRT log 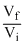
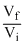
B.W = - nRT log 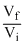
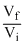
C.W = nRT log 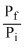
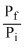
D.W = - nRT log 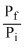
Solution
The work done in expansion of gas
W = nRT l n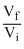 = nRT l n
= nRT l n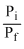
The work done on the gas
= - nRT l n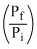
W = nRT l n
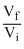 = nRT l n
= nRT l n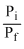
The work done on the gas
= - nRT l n
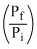
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
A monoatomic gas at pressure P1 and volume V1 is compressed adiabatically to of its original volume. What is the final p...In a process, 500 calories of heat is given to a system and at the same time, 100 joules of work is done on the system. ...A Carnot engine works between temperature 727oC and 27oC. The efficiency of the engine is :...A quantity of air (γ = 1.4) at 27oC is compressed suddenly, the temperature of the air system will :...A gas at intial temperature 27oC is adiabatic cally compressed so that its volume becomes 1 / 9 of the previous one. If ...