ThermodynamicsHard
Question
An ideal gas at 27oC is compressed adiabatically to 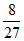 its original volume [TVγ-1] and
its original volume [TVγ-1] and 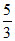 , Then the rise in temprature will be :
, Then the rise in temprature will be :
Options
A.480oC
B.450oC
C.375oC
D.225oC
Solution
Applying the formula
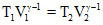
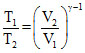
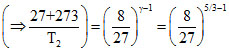
or,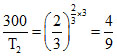
T2 =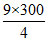 = 675oK
= 675oK
toC = 675 - 273 = 402oC
Rise temperture = 402 - 27 = 375oC
or,
T2 =
toC = 675 - 273 = 402oC
Rise temperture = 402 - 27 = 375oC
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
100 mole of an ideal monoatomic gas undergoes the following thermodynamic process as shown in the figure (P - V or Press...The colour of a star indicates its :...Two moles of a monoatomic ideal gas is confined in a container and is heated such that its temperature increases by 10oC...The surface temperature of a body is 727oC and that of another body is 327oC. The ratio of total energy radiated by them...If the coefficient of Linear expansion of a solid is 0.00009. Its coefficient of volume expansion is :...