AIPMT | 2014ThermodynamicsHard
Question
A monoatomic gas at a pressure P, having a volume V expands isothermally to a volume 2 V and then adiabatically to a volume 16 V. The final pressure ofthe gas is (take γ = 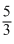 )
)
 )
)Options
A.64 P
B.32 P
C.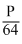
D.16 P
Solution
Step - 1 Isothermal Expansion
PV = P22V ⇒ P2 =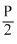
Step - 2 Adiabatic Expansion
P2V2γ = P3V3γ
⇒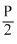 (2V)5/3 = P3(16V)5/3
(2V)5/3 = P3(16V)5/3
⇒ P3 =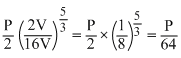
PV = P22V ⇒ P2 =
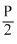
Step - 2 Adiabatic Expansion
P2V2γ = P3V3γ
⇒
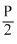 (2V)5/3 = P3(16V)5/3
(2V)5/3 = P3(16V)5/3⇒ P3 =
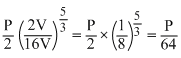
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
If ᐃU and ᐃW represented the increases in internal energy and work done by the system respectively in a ther...One mole of an ideal gas follows the process P = , P0 and V0 are constants when the volume of gas is changed from V0 to ...The temperature of 5 moles of a gas which was held at constant volume was changed from 100oC to 120oC. The change in int...In a water fall the water falls from a height of 100 m. If the entire energy of water is converted into heat. The rise i...A gas mixture contain 1 gm. H2 and 1 gm. He if temp. of gas mixture increase 0oC to 100o C at isobaric process. Than fin...