Chemical EquilibriumHard
Question
Listed in the table are forward and reverse rate constant for the reaction
2NO(g) ⇋ N2(g) + O2(g)
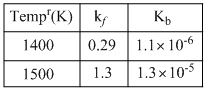
Select the correct statement ?
2NO(g) ⇋ N2(g) + O2(g)

Select the correct statement ?
Options
A.Reaction is exothermic and value of equilibrium constant (Keq) at 1400 K is 3.79 × 10-6
B.Reaction is endothermic and value of Keq at 1400 K is 2.63 × 105
C.Reaction is exothermic and value of Keq at 1400 K is 2.63 × 105
D.Reaction is endothermic and value of Keq at 1500 K is 9.28 × 104.
More Chemical Equilibrium Questions
For the reaction N2O4(g)$\rightleftharpoons$ 2NO2(g), the relation between the degree of dissociation of N2O4(g) at pres...In a closed system : A(s) ⇋ 2B(g) + 3C(g), if the partial pressure of C is doubled at equilibrium, then partial pr...For the reaction X ⇋ 2Y and Z ⇋ P + Q occuring at two different pressure P1 and P2, respectively. The ratio ...In the reaction N2(g) + 3H2(g) $\rightleftharpoons$2NH3(g) + Heat. One mole of N2 reacts with three moles of H2. If at e...For a reaction,A → B ; if log10 K(sec-1) = 14 - the Arrheniums parameter and energy of activation for the reaction...