Chemical EquilibriumHard
Question
In a closed system : A(s) ⇋ 2B(g) + 3C(g), if the partial pressure of C is doubled at equilibrium, then partial pressure of B will be :
Options
A.two times the original value
B.one half of its original value
C.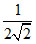 times the original value
times the original value
D.2√2 times the original value
Solution
Kp = p2B(1)p3C(1) = p2B(2)p3C(2) = p2B(2)(2pC(1))3 ;
hence or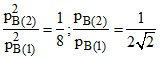
hence or
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
Consider the following reversible reactions (at 298 K) :(a) N2O4 ⇋ 2NO2 (b) 2SO2 + O2 ⇋ 2SO3 (c) 2HI ⇋...Some quantity of water is contained in a container as shown in figure. As neon is added to this system at constant press...The reaction A(g) + B(g) ⇋ C(g) + D(g) is studied in a one litre vessel at 250oC. The initial concentration of A w...1 mole each of H2(g) and I2(g) are introduced in a 1L evacuated vessel at 523K and equilibrium H2(g) + I2(g) ⇋ 2HI...Which of the following is correct for the equilibrium of the reaction C(s) + H2O(g) ⇋ CO(g) + H2(g)...