Chemical EquilibriumHard
Question
For a reaction,
A → B ; if log10 K(sec-1) = 14 -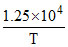 the Arrheniums parameter and energy of activation for the reaction are :
the Arrheniums parameter and energy of activation for the reaction are :
A → B ; if log10 K(sec-1) = 14 -
Options
A.1014 sec-1, 239.34 kJ
B.14,57.6 kcal
C.1014 sec-1, 23.93 kJ
D.1014 sec, 5.76 kcal
Solution
2.303 log10 K = 2.303 log10 A = 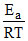
∴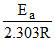 = 1.25 × 104
= 1.25 × 104
∴ Ea = 1.25 × 104 × 2.303 × 8.314
239.34 × 103 J
log A = 14 ∴ A = 1 × 1014
∴
∴ Ea = 1.25 × 104 × 2.303 × 8.314
239.34 × 103 J
log A = 14 ∴ A = 1 × 1014
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
If 0.3 moles of hydrogen gas and 2.0 moles of sulphur solid are heated to 87o C in a 2.0 L vessel, then what will be the...One mole of He (g, colourless) is added to the equilibrium mixture containing N2O4 (g, colourless) and NO2 (g, red-brown...If K1 and K2 are respective equilibrium constants for the two reactions XeF6(g) + H2O(g) ⇋ XeOF4(g) + 2HF(g) XeO4(...In the reaction C(s) + CO2(g) ⇋ 2CO(g) the equilibrium pressure is 12 atm. If 50% of CO2 reacts than Kp will be :...Forty percent of a mixture of 0.2 mol of N2 and 0.6 mol of H2 reacts to give NH3 according to the equationN2(g) + 3H2 (g...