Chemical EquilibriumHard
Question
For the reaction X ⇋ 2Y and Z ⇋ P + Q occuring at two different pressure P1 and P2, respectively. The ratio of the two pressure is 1 : 3. What will be the ratio of equilibrium constant, if the degree of dissociation of X and Z are equal.
Options
A.1 : 36
B.1 : 12
C.1 : 9
D.2 : 3
Solution
for reaction X ⇋ 2Y
Initial 1 0
At equilibrium 1 - α 2α
Total moles = 1 - α + 2α = 1 + α
Px =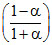 P1; PY =
P1; PY = 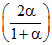 P1
P1
∴ Kp1 =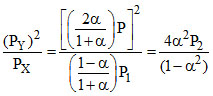 ..... (ii)
..... (ii)
Similarly for X ⇋ P + Q
Initial 1 0 0
At equilibrium 1 - α 2α α
Total moles = 1 - α + α + α = 1 + α
PZ =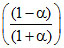 P2; PQ =
P2; PQ = 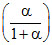 P2; PP =
P2; PP = 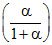
Kp2 =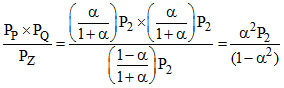 ...... (i)
...... (i)
Dividing equations (i) and (ii), we get,
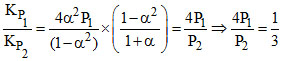 (given)
(given)
∴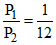
Initial 1 0
At equilibrium 1 - α 2α
Total moles = 1 - α + 2α = 1 + α
Px =
∴ Kp1 =
Similarly for X ⇋ P + Q
Initial 1 0 0
At equilibrium 1 - α 2α α
Total moles = 1 - α + α + α = 1 + α
PZ =
Kp2 =
Dividing equations (i) and (ii), we get,
∴
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
The system PCl5(g) ⇋ PCl3(g) + Cl2(g) attains equilibrium. If the equilibrium cocentrateion of PCl3(g) is doubled,...Solid ammonium carbamate dissociates as: NH2COONH4(s) $\rightleftharpoons$2NH3(g) + CO3(g). In a closed vessel, solid am...Consider the formation of SO3 according to the reaction 2SO2(g) + O2(g) $\rightleftharpoons$2SO3(g); ΔH = −198 kJ.Which ...One mole each of A and B and 3 moles each of C and D are placed in 1 L flask. If equilibrium constant is 2.25 for the re...For a reversible reaction aA + bB ⇋ cC + dD ; the variation of K with temperature is given by log then...