KTGHard
Question
O2 gas is filled up in a cylinder. If pressure is increased 2 times temperature becomes four times. then how much times their density will be :
Options
A.2
B.4
C.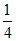
D.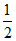
More KTG Questions
The average energy of the molecules of a monoatomic gas at temperature T is :- (K = Boltzmann constant )...The molar heat capacity C for an ideal gas going through a process is given by C = , where ′a′ is a constant...In a process the pressure of a gas is inversely proportional to the square of the volume. If temperature of the gas incr...A gas has :...A cyclic process of an ideal monoatomic gas is shown in figure. The correct statement is (are) :...