KTGHard
Question
The molar heat capacity C for an ideal gas going through a process is given by C = 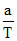 , where ′a′ is a constant. If g =
, where ′a′ is a constant. If g = 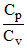 , the work done by one mole of gas during heating from T0 to ηT0 will be:
, the work done by one mole of gas during heating from T0 to ηT0 will be:
Options
A.a ln η
B.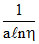
C.alnη − 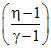 RT0
RT0
D.alnη − (g − 1)RT0
Solution
(C)
dW = dQ − dU
dW = nCdT − nCVdT
W =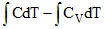
=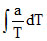 − CV ᐃT
− CV ᐃT
= a ln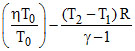
W = a ln η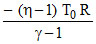
dW = dQ − dU
dW = nCdT − nCVdT
W =
=
= a ln
W = a ln η
Create a free account to view solution
View Solution FreeMore KTG Questions
Which of the following formula is wrong -...The Energy of Monoatomic gas is :-...Consider two boxes containing ideal gases A and B such that their temperatures, pressures and number densities are same....Mole of H2 at 47oC and at pressure 4 atmosphere is mixed with 2 mole of N2 at 127o and at 5 atmosphere such that the vol...The root mean square velocity of hydrogen molecules at 300 K is 1930 m/s. Then the r.m.s. velocity of oxygen molecules a...