KTGHard
Question
In a process the pressure of a gas is inversely proportional to the square of the volume. If temperature of the gas increases, then work done by the gas:
Options
A.is positive
B.is negative
C.is zero
D.may be positive
Solution
P α 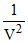 ⇒ P =
⇒ P = 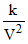 ⇒ PV2 k
⇒ PV2 k
⇒ PV. V = k ⇒ nRTV = k ⇒ TV = k1
Since temperature increases therefore volume decreases or work done by the gas is negative.
⇒ PV. V = k ⇒ nRTV = k ⇒ TV = k1
Since temperature increases therefore volume decreases or work done by the gas is negative.
Create a free account to view solution
View Solution FreeMore KTG Questions
In figure, P-V curve of an ideal gas is given. During the process, the cumulative work done by the gas...The expansion of an ideal gas of mass m in volume at a constant pressure P is given by the straight line B. Then the exp...Pi, Vi are initial pressure and volumes and Vf is final volume of a gas in a thermodynamic process respectively. If PVn ...Consider a gas with density ρ and as the root mean square velocity of its molecules contained in a volume. If the s...An ideal gas is allowed to expand freely against a vacuum in a rigid insulated container. The gas undergoes:...