KTGHard
Question
If K is Boltzman′s constant, m the mass of one molecule and temperature of gas is T, then root mean square velocity will be :-
Options
A.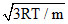
B.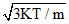
C.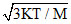
D.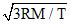
More KTG Questions
Pick the correct statement (s) :...For an ideal gas, the heat capacity at constant pressure is larger than than that at constant volume because...Two containers A and B contain molecular gas at same temperature with mases of melecules are mA and mB, then relation of...The internal energy of an ideal gas is equal to negative of the work done by the system, then...The average speed of nitrogen molecules in a gas is v. If the temperature is doubled and the N2 molecule dissociate into...