KTGHard
Question
Two containers A and B contain molecular gas at same temperature with mases of melecules are mA and mB, then relation of momentum PA and PB will be-
Options
A.PA = PB
B.PA = 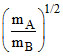 PB
PB
C.PA = 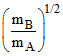 PB
PB
D.PA = 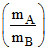 PB
PB
More KTG Questions
The root mean square speed of hydrogen molecules of an ideal hydrogen gas kept in a gas chamber at 0oC is 3180 m/s. The ...Which of the following formula is wrong -...The temperature of ideal gas is the result of which of the following energies :...Two containers of same volume are filled with atomic Hydrogen and Helium respectively at 1 and 2 atm pressure. If the te...In a cyclic process shown on the P − V diagram the magnitude of the work done is :...