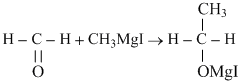Ionic EquilibriumHard
Question
HCHO reacts with CH3MgI followed by hydrolysis to give :-
Options
A.CH3OH
B.CH3CH2OH
C.CH3I
D.CH3CH2I
More Ionic Equilibrium Questions
The pH of solution containing 0.10 M sodium acetate and 0.03 M acetic acid is(pKa for CH3COOH = 4.57)...If pH of a saturated solution of Ba(OH)2 is 12, the value of its Ksp is...For which of the following solutions must we consider the ionisation of water when calculating the pH or pOH :-...Which of following can act as buffer ?...What is [H+] in mol/L of a solution that is 0.20 M in CH3COONa and 0.10 M in CH3COOH ? (Ka for CH3COOH = 1.8 × 10-5...