Ionic EquilibriumHard
Question
If pH of a saturated solution of Ba(OH)2 is 12, the value of its Ksp is
Options
A.4.00 × 10-6 M3
B.4.00 × 10-7 M3
C.4.00 × 10-6 M3
D.4.00 × 10-7 M3
Solution
Given, pH of Ba(OH)2 = 12
∴ [H+]= [1 × 10-12]
and [OH-] =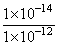 mol
mol
[∵[H+][OH-] = 1×10-14]
= 1×10-2 mol/L
Ba(OH)2 →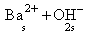
Ksp = [Ba2+][OH-]2
= [s][2s]2
=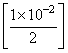 (1 × 10-2)2
(1 × 10-2)2
= 0.5 × 10-6
= 5 × 10-7 (M/lit)3
∴ [H+]= [1 × 10-12]
and [OH-] =
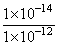 mol
mol[∵[H+][OH-] = 1×10-14]
= 1×10-2 mol/L
Ba(OH)2 →
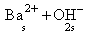
Ksp = [Ba2+][OH-]2
= [s][2s]2
=
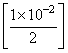 (1 × 10-2)2
(1 × 10-2)2 = 0.5 × 10-6
= 5 × 10-7 (M/lit)3
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
For a sparingly soluble salt ApBq, the relationship of its solubility product (LS) with its solubility (S) is...At 40o C, the density of heavy water is 1.02 g/ml and its ionic product is 5.1 × 10−15. Which of the following if the on...When equal volumes of the following solutions are mixed, precipitation of AgCl (Ksp = 1.8 × 10-1) will occur only w...The pH of 0.1 M – N2H4 solution is (For N2H4, Kb1 = 3.6 × 10−6, Kb2 = 6.4 × 10−12, log 2 = 0.3, log 3 = 0.48)...An amount of 0.01 moles of solid AgCN is rendered soluble in 1 L by adding just sufficient excess cyanide ion to form Ag...