SolutionHard
Question
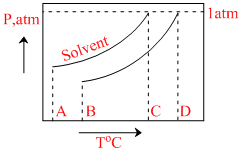
What is the normal boiling point of the solution represented by the phase diagram ?

Options
A.A
B.B
C.C
D.D
Solution
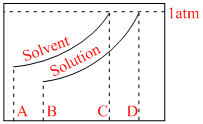
Normal boiling point of the solution is that temperature at which vapour pressure of solution equals to 1 atm.
Create a free account to view solution
View Solution FreeMore Solution Questions
Freezing point lowering expression is ᐃTf = Kfm (molality)Which of the following assumptions are considered for th...The concentration of pollutant in ppm (w/w), that has been measured at 450 mg per 150 kg of sample is :...The vapour pressure of a solution of a non-volatile solute B in a solvent A is 95% of the vapour pressure of the solvent...The solubility of solid silver chromate, Ag2CrO4 is determined in three solvents. (Ksp = 9 × 10-12)(i) Pure water (...The normal freezing point of nitrobenzene is 278.82 K. A non-volatile solute is dissolved in it and a solution of molali...