SolutionHard
Question
When 20 g of naphthoic acid (C11H8O2) is dissolved in 50 g of benzene (Kf = 1.72 K kg mol-1), a freezing point depression of 2 K is observed. The van′t Hoff factor (i) is
Options
A.0.5
B.1
C.2
D.3
Solution
ᐃTf = Kf × molality × i
2 = 1.72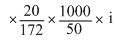
i = 0.5
Hence (A) is correct.
2 = 1.72
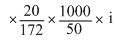
i = 0.5
Hence (A) is correct.
Create a free account to view solution
View Solution FreeMore Solution Questions
A mixture of ethyl alcohol and propyl alcohol has a vapour pressure of 290 mm at 300 K. The vapour pressure of propyl al...At higher altitudes, water boils at temperature < 100oC because...Name the gas that can readily decolourise acidified KMnO4 solution :...Which one of the statements given below concerning properties of solutions, describes a colligative effect ?...Conductivity (unit Siemen′s S) is directly proportional to area of the vessel and the concentration of the solutio...