Chemical Kinetics and Nuclear ChemistryHard
Question
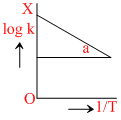
raph between log k and 1/T (k is rate constant in s-1 and T is the temperature in k) is a straight line if OX = 5 and slope of the line = - 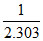 then Ea is :-
then Ea is :-

Options
A.2.303 × 2 cal
B.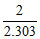 cal
cal
C.2 cal
D.None of these
Solution
log K = log A - 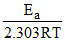
and given slope = -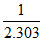 (slope =
(slope = 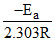 )
)
and given slope = -
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Zn + 2 H+ → Zn2+ + H2Half-life period is independent of concentration of zinc at constant pH. For the constant con...The bromination of acetone that occurs in acid solution is represented by CH3COCH3 (aq) + Br2 (aq) → CH3COCH2 Br (...A radioactive substance ′A′ decay to stable product ′B′ as follow - A → B ;After what time...A certain reaction A → B follows the given concentration (Molarity)-time graph. Which of the following statements ...For the first-order reaction A(g) → 2B(g) + C(g), the total pressure after time t from the start of reaction with A is P...