Chemical Kinetics and Nuclear ChemistryHard
Question
A certain reaction A → B follows the given concentration (Molarity)-time graph. Which of the following statements is/are true ?
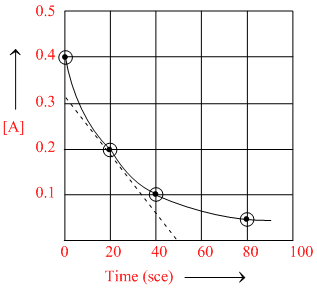

Options
A.The reaction is second order with respect to A.
B.The rate for this reaction at 20 s will be 7 × 10-3 M s-1.
C.The rate for this reaction at 80 s will be 1.75 × 10-3 M s-1.
D.The [B] will be 0.35 M at t = 60 s.
Solution
Use initial rate law method, let the reaction be first order,
So, k =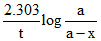
B. At t = 20 s, k1 =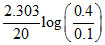 = k1
= k1
⇒ Assumption is correct (k1 = k2)
Rate at 20 s = k[A] =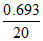 × 0.2
× 0.2
= 0.0063 ≈ 7 × 10-3 M s-1
Clearly, half life t1/2 = 20 s
D. In 60 s, number of life =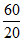 = 3
= 3
⇒ [B] at 60 s
= 0.4 - 0.4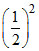 = 0.35 M
= 0.35 M
So, k =
B. At t = 20 s, k1 =
⇒ Assumption is correct (k1 = k2)
Rate at 20 s = k[A] =
= 0.0063 ≈ 7 × 10-3 M s-1
Clearly, half life t1/2 = 20 s
D. In 60 s, number of life =
⇒ [B] at 60 s
= 0.4 - 0.4
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
A reaction involving two different reactants can never be...The rate constant of a reaction is given by k = 2.1 × 1010 exp(-2700/RT). It means that...For The Reaction, N2O5(g) → 2NO2(g) + 1/2 O2(g) the value of rate of disappearance of N2O5 is given as 6.25 ×...Arrange the following in order of decreasing boiling point :-(I) n-butane (II) n-butanol (III) n-butyl chloride (IV) Iso...95Am241 and 90Th234 belong respectively to :...