Chemical EquilibriumHard
Question
Consider the following equilibrium in a closed container N2O4(g) ⇋ 2NO2(g) At a fixed temperature, the volume of the reaction container is halved. For this change, which of the following statements hold teue regarding the equilibrium constant (Kp) and degree of dissociation (α) ?
Options
A.Neither Kp nor α changes
B.Both Kp and α changes
C.Kp changes but α does not change
D.Kp does not change but α changes
Solution
For the reaction; N2O4(g) ⇋ 2NO2(g)
KC =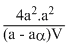 ........(1)
........(1)
where a is initial mole of N2O4 in V litre vessel and α is degree of dissociation of N2O4. If volume is reduced to V/2 , the initial concentration of N2O4 becomes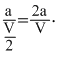
An increase in concentration of reactants leads to foward reaction i. e., the decomposition of N2O4 to have constant value of Kp.
KC =
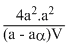 ........(1)
........(1)where a is initial mole of N2O4 in V litre vessel and α is degree of dissociation of N2O4. If volume is reduced to V/2 , the initial concentration of N2O4 becomes
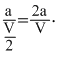
An increase in concentration of reactants leads to foward reaction i. e., the decomposition of N2O4 to have constant value of Kp.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
A 20 litre container at 400 K contains CO2(g) at pressure 0.4 atm and an excess of SrO (neglect the volume of solid SrO)...For a reversible reaction $A\overset{\quad K_{1}K_{2}\quad}{\leftleftarrows}B$, the initial molar concentration of A and...The following equilibria are given : N2 + 3H2 ⇋ 2NH3 K1 N2 + O2 ⇋ 2NO K2 H2 + O2 ⇋ H2O K3 The equilibr...The equilibrium constant for, 2H2S (g) ⇋ 2H2(g) + S2(g) is 0.0118 at 1300 K while the heat of dissociation is 597....Steam decomposes at high temperature according to the following equation.2H2O(g) $\rightleftharpoons$2H2(g) + O2(g); ΔHo...