Chemical EquilibriumHard
Question
The following equilibria are given :
N2 + 3H2 ⇋ 2NH3 K1
N2 + O2 ⇋ 2NO K2
H2 +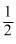 O2 ⇋ H2O K3
O2 ⇋ H2O K3
The equilibrium cinstant of the reaction
2NH3 +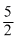 O2 ⇋ 2NO + 3H2O in terms of K1, K2 and K3
O2 ⇋ 2NO + 3H2O in terms of K1, K2 and K3
N2 + 3H2 ⇋ 2NH3 K1
N2 + O2 ⇋ 2NO K2
H2 +
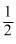 O2 ⇋ H2O K3
O2 ⇋ H2O K3The equilibrium cinstant of the reaction
2NH3 +
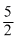 O2 ⇋ 2NO + 3H2O in terms of K1, K2 and K3
O2 ⇋ 2NO + 3H2O in terms of K1, K2 and K3 Options
A.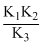
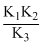
B.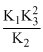
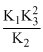
C.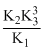
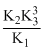
D.K1K2K3
Solution
(I) N2 + O2 ⇋ 2NOH3
K1 =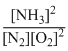
(II)N2 + O2 ⇋ 2NO;
K2 =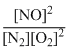
(III) H2 + 1/2O2 ⇋ H2O;
K3 =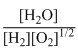
2HN2 + 5/2 O2 ⇋ 2NO + 3H2O;
Kc =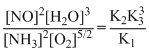
K1 =
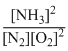
(II)N2 + O2 ⇋ 2NO;
K2 =
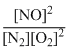
(III) H2 + 1/2O2 ⇋ H2O;
K3 =
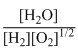
2HN2 + 5/2 O2 ⇋ 2NO + 3H2O;
Kc =
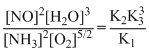
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
At total pressure P1 atm and P2 atm, N2O4 is dissociated to an extent of 33.33% and 50.00%, respectively. The ratio of p...X2(g) + Y2(g) ⇋ 2XY(g) reaction was studied at a certain temperature. In the beginning 1 mole of X2 was taken in a...The condition suitable for forming atomic chlorine from molecular chlorine is...Consider the equilibrium HgO(s) + 4I– (aq) + H2O(l) $\rightleftharpoons$HgI42–(aq) + 2OH– (aq), which changes will decre...KMnO4 can be prepared from K2MnO4 as per the reaction :- 3MnO42- + 2H2O ⇋ 2MnO4- + MnO4 + 4OH-The reaction can go ...