Chemical EquilibriumHard
Question
The equilibrium constant for, 2H2S (g) ⇋ 2H2(g) + S2(g) is 0.0118 at 1300 K while the heat of dissociation is 597.4 kJ. The standard equilibrium constant of the reaction at 1200 K is :
Options
A.1.180 × 10-4
B.11.80
C.118.0
D.cannot be calculated from given data
Solution
log 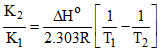
log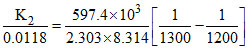
log K2 = - 2 + log (0.0118) = - 3.928 ⇒ K2 = 1.18 × 10-4
Therefore, (A) option is correct.
log
log K2 = - 2 + log (0.0118) = - 3.928 ⇒ K2 = 1.18 × 10-4
Therefore, (A) option is correct.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
Ammonia gas at 15 atm is introduced in a rigid vessel at 300 K. At equilibrium the total pressure of the vessel is found...When $\text{NaNO}_3$ is heated in a closed vessel, oxygen is liberated and $\text{NaNO}_2$ is left behind. At equilibriu...For the chemical reaction 3X(g) + Y(g) ⇋ X3Y(g) The amount X3Y at equilibrium is affected by :...If the value of an equilibrium constant for a particular reaction is 1.6 × 1012, then at equilibrium the system wil...X2(g) + Y2(g) ⇋ 2XY(g) reaction was studied at a certain temperature. In the beginning 1 mole of X2 was taken in a...