Mole ConceptHard
Question
Consider the following statements
1. If all the reactants are not taken in their stoichiometric ratio, then at least one reactant will be left behind.
2. 2 moles of H2(g) and 3 moles of O2(g) produce 2 moles of water.
3. equal wt. of carbon and oxygen are taken to produce CO2 then O2 is limiting reagent.
The above statements 1, 2, 3 respectively are (T = True, F = False)
1. If all the reactants are not taken in their stoichiometric ratio, then at least one reactant will be left behind.
2. 2 moles of H2(g) and 3 moles of O2(g) produce 2 moles of water.
3. equal wt. of carbon and oxygen are taken to produce CO2 then O2 is limiting reagent.
The above statements 1, 2, 3 respectively are (T = True, F = False)
Options
A.T T T
B.F T F
C.F F F
D.T F T
Solution
(1) It is a fact.
(2) 2H2 + O2 → 2H2O
Initial mole 2 3 0
final mole 0 3-1 = 2 2
(3) C + O 2→ CO2
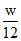
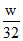
Here C is limiting reagent.
(2) 2H2 + O2 → 2H2O
Initial mole 2 3 0
final mole 0 3-1 = 2 2
(3) C + O 2→ CO2
Here C is limiting reagent.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
In a binary ideal liquid solution of A and B in which vapour pressure of pure component A and B are 600 and 400 torr res...Two bulbs A and B contains 16 g O2 and 16g O3, respectively. Which of the statements are true ?...In which molecule all X - O bond length present in molecule are equal (X = central atom) :-...Which of the following has same mass...If we consider that , in place of ; mass of carbon atom is taken to be the relative atomic mass unit, the mass of one mo...