Mole ConceptHard
Question
Dissolving 120 g of urea (mol. wt. 60) in 1000 g of water gave a solution of density 1.15 g/mL.
The molarity of the solution is
The molarity of the solution is
Options
A.1.78 M
B.2.00 M
C.2.05 M
D.2.22 M
Solution
Total mass of solution = 1000 + 120 = 1120 g
Total volume of solution in (L)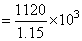
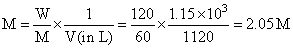
Total volume of solution in (L)
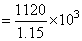
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
An organic compound "P" of molecular formula gives positive lodoform test but negative Tollen's test. When "P" is treate...An amount of 0.3 mole of SrCl2 is mixed with 0.2 mole of K3PO4. The maximum moles of KCl which may form is...When a hydrocarbon is burnt completely, the ratio of masses of CO2 and H2O formed is 44: 27. The hydrocarbon is...The absolute configuration of the two chiral centres in the following molecule are...Out of 1.0 g dioxygen, 1.0 g atomic oxygen and 1.0 g ozone, the maximum number of oxygen atoms are contained in...