ElectrochemistryHard
Question
The standard reduction potential of Cu2+ / Cu and Cu2+ / Cu+ are 0.339V and 0.153 V respectively. The standard electrode potential of Cu+ / Cu half- cell is :
Options
A.0.184 V
B.0.827 V
C.0.525 V
D.0.490 V
Solution
Cu2+ + 2e → Cu ; - ᐃGo1
Cu2+ + e → Cu+ ; - ᐃGo2

Half recaction Cu+ + e → Cu ; - ᐃGo3 = - ᐃGo1 + ᐃGo2
or n × Eo3F = n1 Eo1 F - n2 Eo2 F
Eo3 =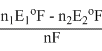
=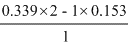 = 0.525V
= 0.525V
Cu2+ + e → Cu+ ; - ᐃGo2

Half recaction Cu+ + e → Cu ; - ᐃGo3 = - ᐃGo1 + ᐃGo2
or n × Eo3F = n1 Eo1 F - n2 Eo2 F
Eo3 =
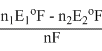
=
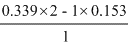 = 0.525V
= 0.525VCreate a free account to view solution
View Solution FreeMore Electrochemistry Questions
Variation of molar conductance of an electrolytic solution with temperature is that it...A hydrogen electrode placed in a buffer solution of CH3COONa and CH3COOH in the ratios of x : y and y : x has electrode ...The distance between two electrodes of a cell is 2.5 cm and area of each electrode is 5 cm2. The cell constant is...The standard reduction potentials Eo, for the half reactions are as Zn → Zn2+ + 2e-, Eo = + 0.76V Fe → Fe2+ ...Pt | (H2) | pH = 1 || pH = 2 | (H2)Pt 1 atm 1 atmThe cell reaction for the given cell is :...