ElectrochemistryHard
Question
Pt | (H2) | pH = 1 || pH = 2 | (H2)Pt
1 atm 1 atm
The cell reaction for the given cell is :
1 atm 1 atm
The cell reaction for the given cell is :
Options
A.spontaneous
B.non - spontaneous
C.equilibrium
D.none of these
Solution
H2 → 2H+(10-2M) + 2e-
2H+(10-3 M) + 2e- → H2
2H+(10-3M) → 2H+(10-2M)
E = 0 -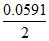 log
log 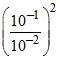
E → - ve (Non spontaneaous).
2H+(10-3 M) + 2e- → H2
2H+(10-3M) → 2H+(10-2M)
E = 0 -
E → - ve (Non spontaneaous).
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
To a beaker containing 0.1 M – HCl, a little pure solid AgCl is added. Both a silver and a hydrogen electrode (PH2 = 1.0...Identification of anode and cathode in an electrochemical cell is made by the use of...On electrolysis, in which of the following, O2 would be liberated at the anode ?...For the electrochemical cell, M|M+||X−|X, $E_{M^{+}|M}^{o}$= 0.44 V and $E_{X|X^{-}}^{o}$= 0.33 V. From this data, one c...Consider the reaction : (T = 298 K)Cl2(g) + 2Br-(aq) → 2Cl- (aq) + Br2 (aq.)The emf of the cell, when [Cl-] = [Br2...