ElectrochemistryHard
Question
A hydrogen electrode placed in a buffer solution of CH3COONa and CH3COOH in the ratios of x : y and y : x has electrode potential values E1 volts and E2 volts, respectively at 25oC. The pKa values of acetic acid is (E1 and E2 are oxidation potentials)
Options
A.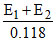
B.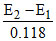
C.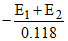
D.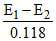
Solution
E1 = 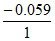 log [H+]
log [H+]
or pH1 = E1 / 0.059 = pKa + log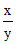
pH2 = E2 / 0.059 = pKa + log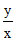
or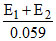 = 2 pKa or pKa =
= 2 pKa or pKa = 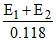
or pH1 = E1 / 0.059 = pKa + log
pH2 = E2 / 0.059 = pKa + log
or
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The oxidation potentials of Zn, Cu, Ag, H2 and Ni are 0.76, -0.34, -0.80, 0.00, 0.25 volt, respectively. Which of the fo...The chemical reaction2AgCl(s) + H2 (g) → 2HCl (aq) + 2Ag (s)taking place in a galvanic cell is represented by the ...In the electrolysis of aqueous CuBr2 using Pt electrodes :...The emf of the cell, Ni | Ni2+ (1.0 M) || Ag+ (1.0M) | Ag [Eo for Ni2+ / Ni = - 0.25 volt, Eo for Ag+/Ag = 0.80 volt] is...The equivalent conductivity of KCl at infinite dilution is 130 S cm2 eq-1. The transport number of Cl-1 ion in KCl at th...