Mole ConceptHard
Question
1M 100 ml NaCl is mixed with 3M 100 ml HCl solution and 1M 200 ml CaCl2 solution. Find the ratio of the concentration of cation and anion.
Options
A.The ratio of concentration of cation and anion = 3/4
B.The ratio of concentration of cation and anion = 2
C.[Cl-] = 2 M
D.[Cl-] = 3/2 M
Solution
Cation concentration = 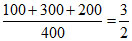 M
M
anion concentration =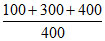
ratio = 3/4
[Cl-] = 2 M
anion concentration =
ratio = 3/4
[Cl-] = 2 M
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Iodobenzene is prepared from aniline (C6H5NH2) in a two-step process as shown here.C6H5NH2+HNO2+HCl?C6H5N2Cl+2H2OC6H5N2C...2KI + I2 + 22 HNO3 → 2HIO3 + 2KIO3 + 22NO2 + 10H2OIf 3 mole of KI & 2 moles I2 are reacted with excess of HNO3. Vo...The mole fraction of NaCl in aqueous solution is 0.2. The solution is...How much NaNO3 must be weighed out to make 50 ml of an aqueous solution containing 70 mg of Na+ per mL ?...A quantity of 10 g of a piece of marble was put into excess of dilute HCl acid. When the reaction was complete, 1120 cm3...