Mole ConceptHard
Question
How much NaNO3 must be weighed out to make 50 ml of an aqueous solution containing 70 mg of Na+ per mL ?
Options
A.12.394 g
B.1.29 g
C.10.934 g
D.12.934 g
Solution
Explanation : M. wt. of NaNO3 = 85
70 mg of Na+ are present in 1 mL
50 ml of solution contains 50 × 70 = 3500 mg = 3.5 g Na+ ion
23 g of Na+ are present in 85 g of NaNO3
3.5 g of Na+ are present in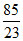 × 3.5 = 12.934 g of NaNO3
× 3.5 = 12.934 g of NaNO3
70 mg of Na+ are present in 1 mL
50 ml of solution contains 50 × 70 = 3500 mg = 3.5 g Na+ ion
23 g of Na+ are present in 85 g of NaNO3
3.5 g of Na+ are present in
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Secondary productivity is rate of formation of new organic matter by :...The reaction of calcium with water is represented by the equation Ca + 2H2O → Ca(OH)2 + H2 What volume of H2 at STP...Vapour pressure of a liquid solution increases -...A sample of ammonium phosphate, (NH4)3PO4, contains 6 moles of hydrogen atoms. The number of moles of oxygen atoms in th...Which of the following is not a characteristic of chemisorption :-(a) Adsorption is irreversible(b) It results into mult...