Mole ConceptHard
Question
The mole fraction of NaCl in aqueous solution is 0.2. The solution is
Options
A.13.9 m
B.Mole fraction of H2O is 0.8
C.acidic in nature
D.neutral
Solution
(A), (B) and (D) Explanation :
(A) xB =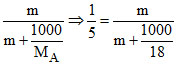 ⇒ m +
⇒ m + 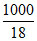 = 5m ⇒ 4m =
= 5m ⇒ 4m = 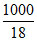 ⇒ m =
⇒ m = 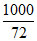 = 13.88
= 13.88
(B) xB = mole fraction of NaCl = 0.2, xNaCl + xH2O = 1, xH2O = 1 - 0.2 = 0.8
(D) is correct because NaCl is salt of NaOH (strong base) and strong acid (HCl).
(C) is not possible.
(A) xB =
(B) xB = mole fraction of NaCl = 0.2, xNaCl + xH2O = 1, xH2O = 1 - 0.2 = 0.8
(D) is correct because NaCl is salt of NaOH (strong base) and strong acid (HCl).
(C) is not possible.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
A molal solution is one that contains one mole of a solute in :...In which of the following reactions, increase in the volume at constant temperature does not affect the number of moles ...When hydrocarbons (alkanes, alkenes or alkynes) are burnt completely in excess of oxygen, then...If ‘JAYARAO BANDA’ is written by a graphite pencil, it weighs 3.0 × 10–10 g. How many carbon atoms are present in it? (N...Which expansion will produce more change in entropy during reversible & isothermal process?...