Mole ConceptHard
Question
0.4 moles of NCl and 0.2 moles of CaCl2 were dissolved in water to have 50 mL of solution, the molarity of Cl ion is :
Options
A.0.8 M
B.1.6 M
C.1.2 M
D.10.0 M
Solution
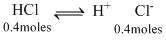
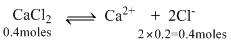
Total Cl- moles - 0.4 + 0.4 = 0.8 moles
Molarity of Cl- =
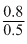 = 1.6 M
= 1.6 MCreate a free account to view solution
View Solution FreeMore Mole Concept Questions
Dissolving 120 g of urea (mol. wt. 60) in 1000 g of water gave a solution of density 1.15 g/mL. The molarity of the solu...Which of the following is not correctly matched :-...A good producer of citric acid is :...To neutralize completely 20 mL of 0.1 M aqueous solution of phosphorous acid (H3PO3), the volume of 0.1 M aqueous KOH so...X and Y are two elements which form X2 Y3 and X3Y4. If 0.20 mol of X2Y3 weighs 32.0 g and 0.4 mol of X3Y4 weighs 92.8 g,...