Mole ConceptHard
Question
Some water is added to a mixture of 250 ml of 0.6 M HCl and 750 ml of 0.2 M HCl to obtain 0.25 M solution of HCl as a resulting solution. Then
Options
A.The volume of water added is 200 ml
B.The volume of water added is 100 ml
C.Total mass of HCl in the solution is 10.95 g
D.Total mass of HCl in the solution is 18.25 g.
Solution
Let V ml water is added
⇒ 0.25 =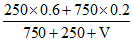
⇒ V = 200 ml
also mass of HCl =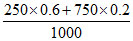 × 36.5 = 10.95 g
× 36.5 = 10.95 g
⇒ 0.25 =
⇒ V = 200 ml
also mass of HCl =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
10 g of CaCO3 contains :-...A mixture of ethylene and excess of H2 had a pressure of 600 mm Hg. The mixture was passed over nickel catalyst to conve...The number of F– ions in 4.2 g AlF3 is (Al = 27, F = 19)...A mixture of N2 and H2 is caused to react in a closed container to form NH3. The reaction ceases before either reactant ...One mole of the complex compound Co(NH3)5Cl3, gives 3 moles of ions on dissolution in water. One mole of the same comple...