Mole ConceptHard
Question
The weight of 1 L of ozonised oxygen at STP was found to be 1.5 g. When 100 mL of this mixture at STP was treated with turpentine oil, the volume was reduced to 90 mL. The molecular weight of ozone is
Options
A.49
B.47
C.46
D.47.9
Solution
Volume of O3 in 100 mL of ozonised O2 = 100 - 90 = 10 mL (dissolved in turpentine)
Volume of O3 in 1 L of ozonised O2 =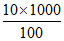 = 100 mL
= 100 mL
Volume of O2 in 1 L = 1000 - 100 = 900 mL
Weight of 900 mL of O2 at STP =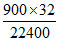 = 1.286 g
= 1.286 g
Weight of 100 mL of O3 at STP = 1.5 - 1.286 = 0.214 g
Now 100 mL of O3 at STP weighs = 0.21 g
for Ozone (0.21/M)×22400= 100
Molecular weight of O3(M) = 47.49
Volume of O3 in 1 L of ozonised O2 =
Volume of O2 in 1 L = 1000 - 100 = 900 mL
Weight of 900 mL of O2 at STP =
Weight of 100 mL of O3 at STP = 1.5 - 1.286 = 0.214 g
Now 100 mL of O3 at STP weighs = 0.21 g
for Ozone (0.21/M)×22400= 100
Molecular weight of O3(M) = 47.49
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
If an iodized salt contains 1% of KI and a person takes 2 g of the salt every day, the iodine ions going into his body e...Which of the following is dependent on temperature?...Equal volume of 0.1M NaCl and 0.1M FeCl2 are mixed with no change in volume due to mixing. Which of the following will b...One mole of mixture of N2, NO2 and N2O4 has a mean molar mass of 55.4 g. On heating to a temperature, at which all the N...A gaseous mixture contains 40% H2 and 60% He by volume. What is the total number of moles of gases present in 10 g of su...