Mole ConceptHard
Question
A 5L vessel contains 2.8 g of N2. When heated to 1800 K, 30% molecules are dissociated into atoms.
Options
A.Total no. of moles in the container will be 0.13
B.Total no. of molecules in the container will be close to 0.421 × 1023.
C.Total no. of moles in the container will be 0.098.
D.All of these are correct.
Solution
(A) and (B) Explanation : 30% of molecule dissociated N2 → 2N
Amount of N2 left =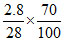 = 0.1 × 0.7 = 0.07
= 0.1 × 0.7 = 0.07
(in moles)
No. of moles of N atoms formed = 2 ×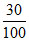 × 0.1 = 0.06
× 0.1 = 0.06
(A) Total no . of moles = 0.07 + 0.06 = 0.13
(B) Total number of molecules = 0.07 × 6.023 × 1023 = 4.2 × 1022 molecule = 0.421 × 1023
∴ We have to calculate molecule of nitrogen not atoms.
Amount of N2 left =
(in moles)
No. of moles of N atoms formed = 2 ×
(A) Total no . of moles = 0.07 + 0.06 = 0.13
(B) Total number of molecules = 0.07 × 6.023 × 1023 = 4.2 × 1022 molecule = 0.421 × 1023
∴ We have to calculate molecule of nitrogen not atoms.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Given below are two statements:Statement I: The number of species among ${SF}_{4}$, ${NH}_{4}^{+},\ \left\lbrack {NiCl}_...Elements P and Q form two types of non-volatile, non-ionizable compounds PQ and . When 1 g of PQ is dissolved in 50 g of...1021 molecules are removed from 200mg of CO2. The moles of CO2 left are :...For the estimation of nitrogen, 1.4 g of an organic compound was digested by Kjeldahl method and the evolved ammonia was...Some bottles of colourless liquids were being labelled when the technicians accidentally mixed them up and lost track of...