Mole ConceptHard
Question
Calculate the mass percent (w/w) of sulphuric acid in a solution prepared by dissolving 4 g of sulphur trioxide in a 100 ml sulphuric acid solution containing 80 mass percent (w/w) of H2SO4 and having a density of 1.96 g/ml. (molecular weight of H2SO4 = 98). Take reaction SO3 + H2O → H2SO4
Options
A.80.8%
B.84%
C.41.65%
D.None of these
Solution
Mass of H2SO4 formed by 4g SO3 = 4.9 g
Mass % of H2 SO4 =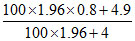 = 80.8 %
= 80.8 %
Mass % of H2 SO4 =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
When burnt in air, a 12.0 g mixture of carbon and sulphur yields a mixture of CO2 and SO2, in which the number of moles ...An amount of 0.15 moles of K2Cr2O7 is required to oxidize a mixture of XO and X2O3 (total mass = 25.56 g) to form XO4 − ...100 gm impure CaCO3 on heating gives 5.6 lt. CO2 gas at STP. Find the percentage of calcium in the lime stone sample. [A...The mass composition of universe may be given as 90% H2 and 10% He. The average molecular mass of universe should be...A volume of 10 ml of gaseous C4Hx exactly requires 55 ml O2 for complete combustion. The value of ‘x’ is...