Mole ConceptHard
Question
100 gm impure CaCO3 on heating gives 5.6 lt. CO2 gas at STP. Find the percentage of calcium in the lime stone sample. [At. wt. Ca = 40 ; C = 12 ; O = 16]
Options
A.10
B.20
C.1
D.30
Solution
CaCO3 → CaO + CO2
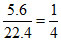 mole
mole
mole of CaO = mole of Ca =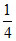
mass of Ca =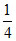 × 40 = 10
× 40 = 10
% of Ca in sample =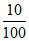 × 100 = 10%
× 100 = 10%
mole of CaO = mole of Ca =
mass of Ca =
% of Ca in sample =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
The volume occupied by 20 g water at 1.2 atm and 4°C is about...H3PO4 (98 g mol-1) is 98% by mass of solution. If the density is 1.8 gm/ml, the molarity is...Specialized cells in the stomach release HCl to aid digestion. If they release too much, the excess can be neutralized b...A quantity of 10 g of a mixture of C2H6 and C5H10 occupy 4480 ml at 1 atm and 273 K. The percentage of C2H6 by mass in t...A power company burns approximately 500 tons of coal per day to produce electricity. If the sulphur content of the coal ...