Mole ConceptHard
Question
When burnt in air, a 12.0 g mixture of carbon and sulphur yields a mixture of CO2 and SO2, in which the number of moles of SO2 is half that of CO2. The mass of the carbon the mixture contains is (At. wt. S = 32)
Options
A.4.08 g
B.5.14 g
C.8.74 g
D.1.54 g
Solution
Let mass of C = X gm
C + O2 → CO2
Initial mole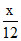 - 0
- 0
final mole 0 2a
S + O2 → SO2
Initial mole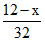 - 0
- 0
final mole 0 - a
Mole of C = Mole of CO2
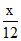 = 2a
= 2a
Mole of S = Mole of SO2
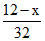 = a
= a
∴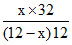 = 2
= 2
x =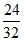 (12 - x)
(12 - x)
4x = 36 - 3x
x =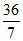 = 5.14 gm
= 5.14 gm
C + O2 → CO2
Initial mole
final mole 0 2a
S + O2 → SO2
Initial mole
final mole 0 - a
Mole of C = Mole of CO2
Mole of S = Mole of SO2
∴
x =
4x = 36 - 3x
x =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Two samples of HCl of 1 M and 0.25 M are mixed. Find volumes of these samples taken in order to prepare 0.75 M HCl solut...How many moles of magnesium phosphate, Mg3(PO4)2 will contain 0.25 mole of oxygen atoms?...On reduction with hydrogen, 3.6 g of an oxide of metal left 3.2g of metal. If the vapour density of metal is 32, the sim...A 10.0 g sample of a mixture of calcium chloride and sodium chloride is treated with Na2CO3 solution. This calcium carbo...H3PO4 (98 g mol-1) is 98% by mass of solution. If the density is 1.8 gm/ml, the molarity is...