Mole ConceptHard
Question
If 0.50 mole of BaCl2 is mixed with 0.20 mole of Na3 PO4, the maximum number of moles of Ba3(PO4)2 that can be formed is :
Options
A.0.70
B.0.50
C.0.20
D.0.10
Solution
3BaCl2 + 2Na3 OP4 → Ba3 (PO4)2 + 6NaCl
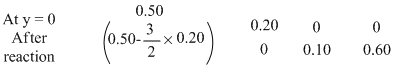
= 0.20
Thus 0.20 mole of Na3 PO4 will give 0.10 mole of Ba3 (PO4)2
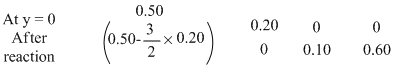
= 0.20
Thus 0.20 mole of Na3 PO4 will give 0.10 mole of Ba3 (PO4)2
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Two elements A and B combine to form compound X and Y. For the fixed mass of A, masses of B combined for the compounds X...The number of hydrogen atoms in 0.9 g glucose, C6H12O6, is same as...When the hydrocarbon propane is burned in air, carbon dioxide and water are formed. If 0.15 mol of CO2 is produced, then...The volume of one mole of water at 277 K is 18 ml. One ml of water contains 20 drops. The number of molecules in one dro...In a textile mill, a double effect evaporator system concentrates weak liquor containing 4% (by mass) caustic soda to pr...