ThermodynamicsHard
Question
The following is (are) endothermic reaction (s) :
Options
A.combustion of methane
B.decomposition of water
C.dehydrogenation of ethane to rthylene
D.conversion of graphite to diamond
Solution
Combustion reactions are always exothermic.
(b) 2H2O → 2H2 + O2
Four σ - bond (O - H) are broken while 3σ (2H - H, 1 O - O) and one π - bond (O = O)is formed, thus energy is absorbed.
(c)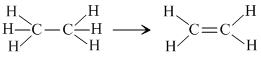
Two σ - bond (C - H) are broken at the cost of one π (C - C) and one σ (H - H) bond.
(d) Diamond has higher energy than graphite
(b) 2H2O → 2H2 + O2
Four σ - bond (O - H) are broken while 3σ (2H - H, 1 O - O) and one π - bond (O = O)is formed, thus energy is absorbed.
(c)
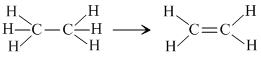
Two σ - bond (C - H) are broken at the cost of one π (C - C) and one σ (H - H) bond.
(d) Diamond has higher energy than graphite
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
In any natural process :-...The enthalpy of vaporization of a liquid is 30 kJ mol-1 and entropy of vaporization is 75 J mol-1. The boiling point of ...For a reaction, A ⇌ P, the plots of [A] and [P] with time at temperatures T1 and T2 are given below. If T2 > T1...1 mol of NH3 gas at 27oC is expanded under adiabatic condition to make volume 8 times (g = 1.33). Final temperature and ...Two moles of an ideal gas compressed isothermally (100oC) and revversibly from a pres sure of 10atm to 25 atm, then the ...