ThermodynamicsHard
Question
Two moles of an ideal gas compressed isothermally (100oC) and revversibly from a pres sure of 10atm to 25 atm, then the free energy cahnge is :
Options
A.+15.482 kJ
B.+10.462kJ
C.+5.684 kJ
D.+3.364 kJ
Solution
For isothermal reversible process,
Wrev = - 2.303 nRT log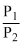
= - 2.303 × 2 × 8.314 × 373 log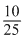
= + 5684.1 J = 5.64 kJ
Wrev = - 2.303 nRT log
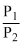
= - 2.303 × 2 × 8.314 × 373 log
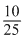
= + 5684.1 J = 5.64 kJ
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
For the reaction of the one mole of zinc dust with one mole of sulphuric acid in a bombcalorimeter, ᐃU and w corre...ΔS will be highest for which of the following reaction?...Which of the following statement(s) is/are false ?...With what minimum pressure must a given volume of an ideal gas ($\gamma$ = 1.4), originally at 400 K and 100 kPa, be adi...Which of the following graphs between pressure 'P' versus volume 'V' represent the maximum work done?...