JEE Advanced | 2018ThermodynamicsHard
Question
For a reaction, A P, the plots of [A] and [P] with time at temperatures T1 and T2 are given below.
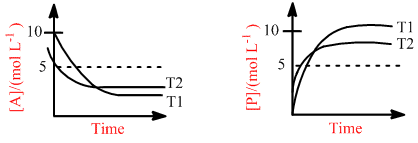
If T2 > T1, the correct statement(s) is (are) (Assume and are independent of temperature and ratio of lnK at T1 to lnK at T2 is greater than . Here H, S, G and K are enthalpy, entropy, Gibbs energy and equilibrium constant, respectively.)
Options
A.
H < 0, S< 0
B.
G < 0, H> 0
C.
G < 0, S < 0
D.
G< 0, S > 0
Solution
A P
given T2 > T1
– > –
(–H° + T1S°) > (–H° + T2 S°)
T1 S° > T2S°
S° < 0
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Identify the intensive quantites from the following :...Equal moles of He, H2, CO2 and SO3 gases are expanded adiabatically and reversibly from the same initial state to the sa...The minimum work which must be done to compress 16 g of oxygen isothermally at 300 K from a pressure of 1.01325 × 103 N/...Inversion temperature is defined as the temperature above which a gas gets warm up and below which, the gas become coole...The molar heat capacity of water in equilibrium with ice at constant pressure is...