Mole ConceptHard
Question
The density of air is 0.001293 g/ml at S.T.P. Its vapour density will be :
Options
A.10
B.15
C.1.44
D.14.4
Solution
Density of air = 0.001293 gm/ml
1 mole of air at STP have 22.4 lit volume
∴ Mass of 1 mole = 22400 × 0.001293 = 28.96 gm
Vapour density =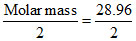 = 14.48.
= 14.48.
1 mole of air at STP have 22.4 lit volume
∴ Mass of 1 mole = 22400 × 0.001293 = 28.96 gm
Vapour density =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
The density of 3M sodium thiosulphate is 1.25 g/ml. Identify the correct statements among the following :...In a textile mill, a double effect evaporator system concentrates weak liquor containing 4% (by mass) caustic soda to pr...A person needs an average of 2.0 mg of riboflavin (vitamin B 2 ) per day. How many grams of butter should be taken by th...A quantity of 1 kg of 1 m glucose solution is diluted to 5 kg. The molality of the diluted solution should be...A student has been given 0.314 g of an organic compound and asked to estimate Sulphur. During the experiment, the studen...