Mole ConceptHard
Question
The density of 3M sodium thiosulphate is 1.25 g/ml. Identify the correct statements among the following :
Options
A.% by weight of sodium thisoulphate is 37.92
B.The mole fraction of sodium thisoulphate is 0.065
C.The molarity of Na+ is 2.53 and S2O32- is 1.25
D.All of these
Solution
(A) and (B) Explanation :
M =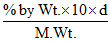 [M. Wt. of Na2S2O3 = 46 + 64 + 48 = 158]
[M. Wt. of Na2S2O3 = 46 + 64 + 48 = 158]
3 =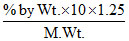
% by wt. =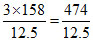 = 37.92 (A) is correct.
= 37.92 (A) is correct.
m =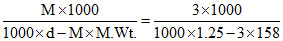
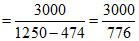 = 3.86
= 3.86
xB =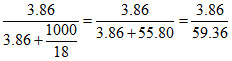 = 0.065 (B) is correct.
= 0.065 (B) is correct.
M =
3 =
% by wt. =
m =
xB =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
An amount of 2 moles of KClO3 is decomposed completely to produce O2 gas. How many moles of butene C4H8 can be burnt com...The fractional abundance of Cl35 in a sample of chlorine containing only Cl35 (atomic weight = 34.9) and Cl37 (atomic we...A quantity of 0.25 g of a substance when vaporized displaced 50 cm3 of air at 0o C and 1 atm. The gram molecular mass of...When 12 g graphite is burnt in sufficient oxygen, CO as well as CO2 is formed. If the product contains 40% CO and 60% CO...2KI + I2 + 22 HNO3 → 2HIO3 + 2KIO3 + 22NO2 + 10H2OIf 3 mole of KI & 2 moles I2 are reacted with excess of HNO3. Vo...