ThermochemistryHard
Question
NH3(g) + 3Cl2(g) → NCl3(g) + 3HCl(g) ; ᐃH1
N2(g) + 3H2(g) → 2NH3(g) ; ᐃH2
H2(g) + Cl2(g) → 2HCl(g) ; ᐃH3
The heat of formation of NCl3(g) in the terms of ᐃH1, ᐃH2 and ᐃH3 is
N2(g) + 3H2(g) → 2NH3(g) ; ᐃH2
H2(g) + Cl2(g) → 2HCl(g) ; ᐃH3
The heat of formation of NCl3(g) in the terms of ᐃH1, ᐃH2 and ᐃH3 is
Options
A.ᐃHf = - ᐃH1 + 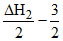 ᐃH3
ᐃH3
B.ᐃHf = ᐃH1 + 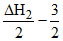 ᐃH3
ᐃH3
C.ᐃHf = ᐃH1 - 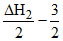 ᐃH3
ᐃH3
D.None
Solution
ᐃH = - ᐃH1 +
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
ΔfHo for NF3(g) is −113 kJ/mol. Bond energy for the N–F bond is 273.5 kJ/mol. The bond energies of N2 and F2, if their m...Enthalpy of neutralization of the reaction between CH3COOH(aq) and NaOH(aq) is −13.2 kcal/eq and that of the reaction be...When 1 g equivalent of strong acid reacts with strong base, the heat released is 13.5 kcal. When 1 g equivalent H2A is c...Among the following, for which reaction the heat of reaction represents bond energy of HCl?...Calculate the enthalpy of formation (in kcal/mol) of HI(g) from the following data.H2(g) + Cl2(g) → 2HCl(g): ΔH = −44.20...