ThermochemistryHard
Question
When 1.0 g of oxalic acid (H2C2O4) is burned in a bomb calorimeter whose heat capacity is 8.75 kJ/K, the temperature increases by 0.312 K. The enthalpy of combustion of oxalic acid at 27oC is :
Options
A.-245.7 kJ/mol
B.-244.452 kJ/mol
C.-246.947 kJ/mol
D.None of these
Solution
H2C2O4(l) + 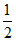 O2(g) → H2O(l) + 2CO2(g);
O2(g) → H2O(l) + 2CO2(g);
ᐃng = 3/2
ᐃUc = -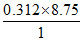 × 90
× 90
= - 245.7 kJ/mol
ᐃH = ᐃU + ᐃngRT
= - 245.7 +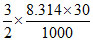 = - 246.947 kJ/mol
= - 246.947 kJ/mol
ᐃng = 3/2
ᐃUc = -
= - 245.7 kJ/mol
ᐃH = ᐃU + ᐃngRT
= - 245.7 +
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
ΔfHo for NF3(g) is −113 kJ/mol. Bond energy for the N–F bond is 273.5 kJ/mol. The bond energies of N2 and F2, if their m...Diborane is a potential rocket fuel which undergoes combustion according to the following reaction.B2H6(g) + 3O2(g) → B2...Determine enthalpy of formation for H2O2 (l), using the listed enthalpies of reactions :N2H4 (l) + 2H2O2 (l) → N2 ...The enthalpy changes for the following processes are listed below: Cl2(g) = 2Cl(g), 242.3 kJ mol-1 I2(g) = 2I(g), 151.0 ...The polymerization of ethylene to linear polyethylene is represented by the reactionnCH2 = CH2→ (−CH2−CH2−)nwhere n has ...