ThermochemistryHard
Question
Determine enthalpy of formation for H2O2 (l), using the listed enthalpies of reactions :
N2H4 (l) + 2H2O2 (l) → N2 (g) + 4H2O (l); ᐃrHo1 = - 818 kJ/mol
N2H4 (l) + O2 (g) → N2 (g) + 2H2O (l); ᐃrHo2 = - 622 kJ/mol
H2 (g) +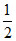 O2 (g) → H2O (l); ᐃrHo3 = - 285 kJ/mol
O2 (g) → H2O (l); ᐃrHo3 = - 285 kJ/mol
N2H4 (l) + 2H2O2 (l) → N2 (g) + 4H2O (l); ᐃrHo1 = - 818 kJ/mol
N2H4 (l) + O2 (g) → N2 (g) + 2H2O (l); ᐃrHo2 = - 622 kJ/mol
H2 (g) +
Options
A.-383 kJ/mol
B.-187 kJ/mol
C.-498 kJ/mol
D.None of these
Solution
For H2(g) + O2(g) → H2O2(l)
DfHo (H2O2,l) = ᐃrH3o +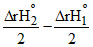
DfHo (H2O2,l) = ᐃrH3o +
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
Calculate the magnitude of resonance energy of CO2 from the following data (in kJ/mol).Bond energies: C=O = 539.0, O=O =...The enthalpy of combustion of propane (C3H8) gas in terms of given data is : Bond energy (kJ/mol) εC-H εO=O &#...Butane exists in various conformations in nature. At any given instant, the probability that a given butane molecule is ...Calculate proton affinity of NH3(g) from the following data.ΔHdissociation H2 = 218 kJ mole–1ΔHdissociation Cl2 = 124 kJ...Oxidising power of chlorine in aqueous solution can be determined by the parameters indicated below: The energy involved...