ThermochemistryHard
Question
The enthalpy changes for the following processes are listed below:
Cl2(g) = 2Cl(g), 242.3 kJ mol-1
I2(g) = 2I(g), 151.0 kJ mol-1
ICl(g) = I(g) + Cl(g), 211.3 kJ mol-1
I2(s) = I2(g), 62.76 kJ mol-1
Given that the standard states for iodine and chlorine are I2(s) and Cl2(g), the standard enthalpy of formation for ICl(g) is
Cl2(g) = 2Cl(g), 242.3 kJ mol-1
I2(g) = 2I(g), 151.0 kJ mol-1
ICl(g) = I(g) + Cl(g), 211.3 kJ mol-1
I2(s) = I2(g), 62.76 kJ mol-1
Given that the standard states for iodine and chlorine are I2(s) and Cl2(g), the standard enthalpy of formation for ICl(g) is
Options
A.-14.6 kJ mol-1
B.-16.8 kJ mol-1
C.+ 16.8 kJ mol-1
D.+ 244.8 kJ mol-1
Solution
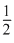 I2(S) +
I2(S) + 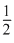 Cl2 → ICl(g)
Cl2 → ICl(g)ᐃH =
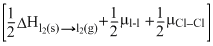 -[μI - Cl]
-[μI - Cl]=
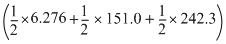 - (211.3)
- (211.3)= 228.03 - 211.3
ᐃH = 16.73
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
A quantity of 1.6 g sample of NH4NO3 is decomposed in a bomb calorimeter. The temperature of the calorimeter decreases b...Match the transformations in column I with appropriate options in column II...The standard heat of formation of carbon atom in gaseous state in kJ/mol is - áƒHo (atomisation of CO gas) = 1072 kJ/mo...For an ionic solid MX2, where X is monovalent, the enthalpy of formation of the solid from M(s) and X2(g) is 1.5 times t...The enthalpy change when x g of phenol dissolves in y g of CHCl3 is given below at a certain temperature.x Y ΔH (kcal)0....